x-linked inheritance
X-linked inheritance means that the faulty gene is located on the X chromosome, the larger of the two sex chromosomes.
Search results
X-linked inheritance means that the faulty gene is located on the X chromosome, the larger of the two sex chromosomes.
Beacon Therapeutics announced positive results from their phase 2 gene therapy trial for X-linked retinitis pigmentosa (XLRP) caused by mutations in the RPGR gene.
It was previously believed that female carriers of X-linked inherited retinal diseases (IRDs) like X-linked retinitis pigmentosa (RP and Choroideremia) remained unaffected by sight loss.
ProQR has published the top-line results from its Phase 2/3 “Illuminate” trial of the RNA therapy sepofarsen for Leber congenital amaurosis type 10 caused by a specific mutation in the CEP290 gene.
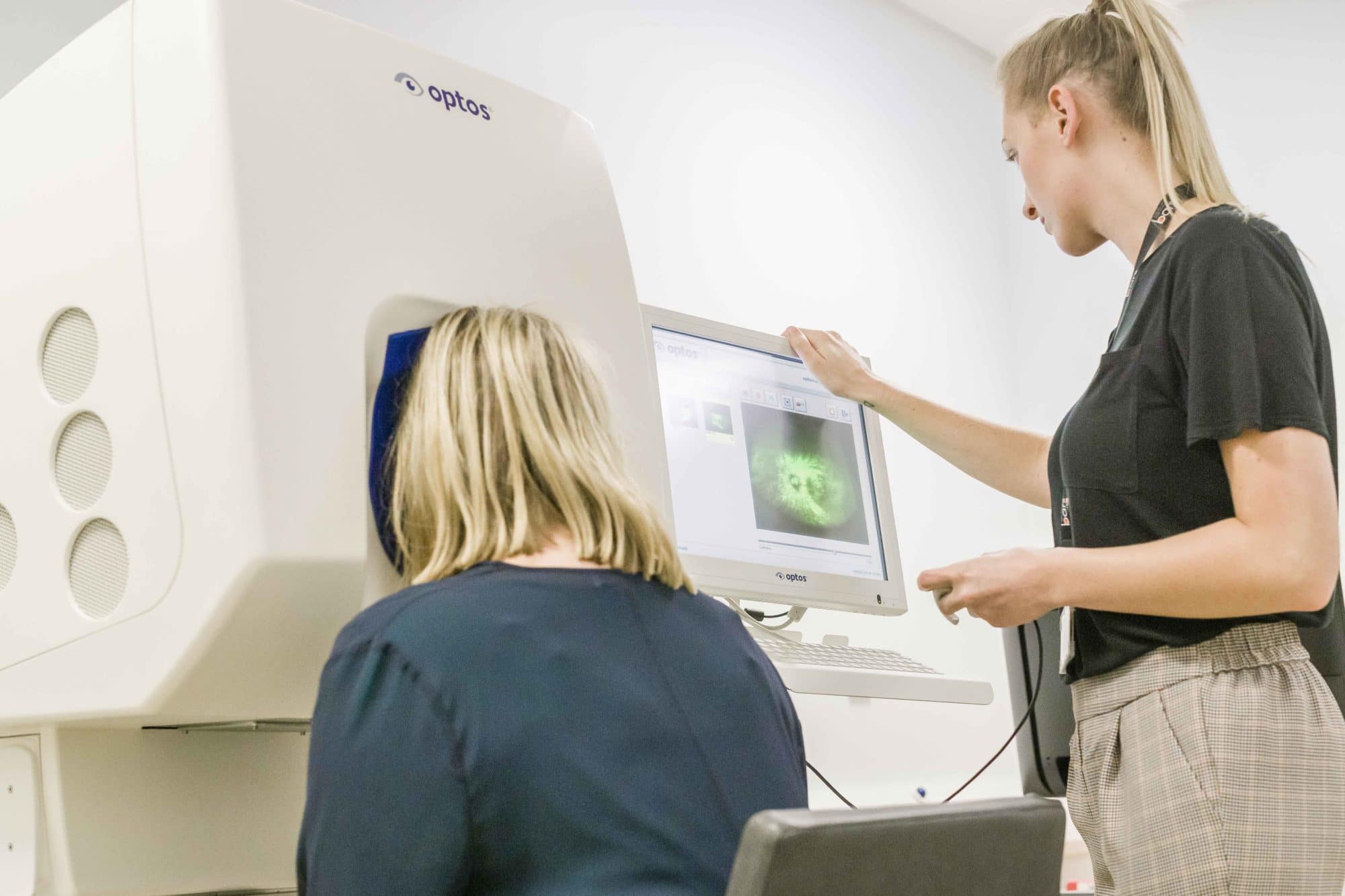
Disulfiram (Antabuse), FDA-approved for deterring alcohol, is in a phase 1 trial at the University of Washington to improve vision in retinitis pigmentosa.
UK researchers have discovered that passing a weak electrical current between electrodes on a person’s scalp may lead to a reduction in frequency of the visual hallucinations experienced by some people living with sight loss.
A round-up of the latest research into inherited retinal conditions - February 2024.
Autosomal dominant inheritance means that the faulty gene is on an autosome (any chromosome other than the sex chromosomes) and that only one faulty copy is needed to cause sight loss.
A recently published study has described numerous disease mechanisms that appear to be common across different types of inherited sight loss, suggesting that there is significant potential for drug treatments that could work regardless of the underlying causative gene.

This edition includes exciting news about our Annual and Professionals’ Conferences. We hope as many of you as possible can join us in Manchester, or online, in September. You’ll also find updates about the latest research and a feature on one of our funded researchers, Dr Jörn Lakowski.